As keepers of the environment, we have a responsibility to preserve, protect, and live harmoniously with the animals that share our planet. As a source of critical proteins that detect the presence of endotoxins in products and instruments used by humans, the Atlantic horseshoe crab has long been treasured by the biopharmaceutical industry. For this reason, it is critical that we serve as advocates for the humane treatment of these animals and strive to achieve balance between our need for this valuable material and the livelihood of the animal that provides it. Over the past 25 years, Charles River Laboratories has worked tirelessly to protect and preserve the long-term health of horseshoe crab populations through horseshoe crab conservation efforts, legislative support, and educational campaigns. In South Carolina, we played an instrumental role in the passage of legislation that banned the use of horseshoe crabs as bait for the eel and whelk fisheries, and as a result, the horseshoe crab populations in South Carolina waterways have flourished. Our horseshoe crab conservation and education efforts have also contributed to a healthy ecosystem for other wildlife.
The biomedical industry’s need for the horseshoe crab has, in fact, driven the development of laws to protect the animal. Their best security is the biomedical industry’s continued reliance on them. Without the need for limulus amebocyte lysate (LAL), the legal protection of the horseshoe crab is not guaranteed, and they would again fall prey to overfishing and be used as bait for eel and whelk. For this reason, it is critical in our horseshoe crab conservation efforts that we serve as advocates for the humane treatment of these animals and strive to achieve balance between our need for this valuable material and the livelihood of the animal that provides it. Thanks to horseshoe crab biomedical research, the horseshoe crab maintains its protective status, allowing the population to continue to flourish.
Limulus polyphemus, known as the Atlantic horseshoe crab, is an ancient mariner with a lineage that dates back
Though they look similar to crustaceans, they actually belong to the subphylum Chelicerata, relating them more closely to spiders and scorpions. Limulus polyphemus is found along the Atlantic and Gulf coasts from northern Maine to the Yucatán Peninsula, with the Delaware Bay as the center of the population. Horseshoe crab conservation is imperative, not only for the pivotal role these animals play in their ecosystems, but also because the blood of the horseshoe crab is vital for developing life-saving therapies.
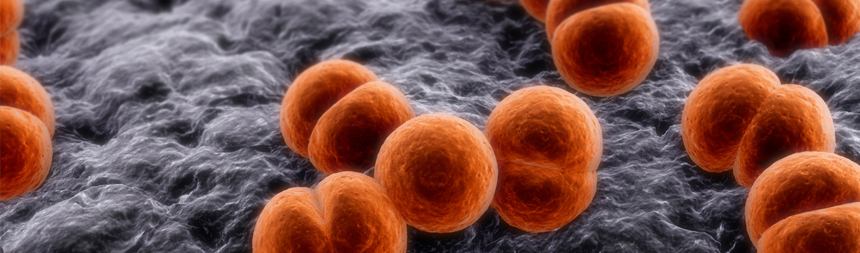
The true beauty of this remarkable creature, however, is seen in its primitive immune system. Instead of developing antibodies in response to infection, the immune system releases proteins that can bind and kill bacteria. In addition, other immune proteins clot when exposed to waterborne bacteria, a mechanism which forms the basis of the LAL test.

Limulus amebocyte lysate (LAL) is an aqueous extract of blood cells (amebocytes) from the horseshoe crab. Comprised of proteins, LAL is used to detect the presence of endotoxins. In the
50 years since this discovery, horseshoe crab blood has served as a natural indicator of bacterial contamination in critical medical treatments all over the world, further making horseshoe crab conservation efforts a top priority.
Why is endotoxin detection so important? Endotoxins are a cell wall component of Gram-negative bacteria that cause a pyrogenic response (fever). They are extremely potent, heat stable, and present everywhere bacteria are or have been. With its specificity, simplicity, and remarkable sensitivity, LAL, which is derived from horseshoe crab blood, ensures the safety of many biopharmaceutical products.
Globally, over 55% of all injectable pharmaceuticals or implantable devices that come in contact with patients’ blood are tested with our LAL products, made possible by horseshoe crab biomedical research. Vaccines, IV drugs, antibiotics, and implantable medical devices are all examples of biopharmaceutical products that need to be tested to ensure they are free of bacterial endotoxin to save and improve the lives of patients.
Learn more about our LAL products. >>
Prior to FDA approval of LAL in the 1980s, the rabbit pyrogen test was the standard FDA-approved test for endotoxins. This test, however, was labor intensive, lengthy, and expensive. Years of research and application have proven LAL to be unequivocally recognized as the most sensitive method available for the detection of endotoxins, making horseshoe crab biomedical research imperative.To illustrate, LAL can detect as little as 1 pg/mL of harmful bacterial endotoxin (equivalent to one grain of sand in an Olympic-sized pool).Approximately 70 million LAL tests are performed each year, and there have been no FDA-confirmed pyrogenic outbreaks due to the failure of LAL detecting the presence of endotoxin. As a highly regulated product, manufactured LAL undergoes biannual inspections by the FDA and must conform to cGMP requirements. Of course, to manufacture LAL, horseshoe crab blood must be carefully collected through a controlled bleeding program, that adheres to horseshoe crab conservation standards. Policies such as hand-harvesting and returning to the sea within 36 hours ensure the animals’ safety. With a focus on horseshoe crab conservation, innovation, humane care and environmental protection, Charles River has responsibly manufactured LAL since 1987.
Learn more about our rapid endotoxin products. >>

Charles River has been a pioneer in the realm of endotoxin detection and horseshoe crab conservation efforts, developing an FDA-licensed LAL cartridge that further safeguards the 440-million-year-old species by using 95 percent less raw material than traditional LAL tests. If all tests were performed using this optimized technology, today’s entire worldwide demand could be met with the horseshoe crab blood collected from our current annual supply of animals. Our cartridge technology is the most environmentally conscious strategy because it significantly reduces the amount of LAL per test and minimizes the need for retesting that is often necessary with traditional methods.
Learn more about our FDA-licensed cartridge technology. >>

In 2006, Charles River launched the Endosafe®-PTS™, the first portable endotoxin test system that accelerates the discovery and development of new drugs by detecting endotoxin contaminants in drugs, medical devices and biological products. Astronauts aboard the International Space Station began using the system to perform biological studies necessary for an extended human presence in space–from crew health and spacecraft environmental studies to the search for life elsewhere in the solar system. NASA has used the Endosafe®-PTS™ to monitor the environment for microbial contamination during the construction of the Mars exploration rovers "Spirit" and "Opportunity". Future applications of PTS and horseshoe crab biomedical research may include monitoring the spread of Earth-derived biological material to the Moon and detecting signs of microbial life on Mars.
Learn more about our adventures in space. >>
In order to produce LAL, we need an extract from the blood of the horseshoe crab. As part of our horseshoe crab conservation efforts, we are committed to obtaining this valuable resource in a humane manner that is both environmentally sustainable and ensures medical treatments are safe for patients around the globe. We only contract with licensed fishermen who are trained to collect horseshoe crabs from the coastal waters. This is a highly controlled and monitored process in which the crabs are carefully and safely collected and then returned to their home waters in less than 36 hours.
Once the crabs are brought into our lab, they are carefully inspected by trained employees who determine the animal’s health and maturity. During this time, the Department of Natural Resources is able to tag and count crabs for population tracking. This opportunity is set up by Charles River as part of our commitment to horseshoe crab conservation. After careful collection of a measured amount of horseshoe crab blood, the crabs are returned, unharmed, to their natural habitat, and their blood volume rebounds quickly. These practices have allowed us to achieve an industry-leading survival rate of donor animals.

Much of the continued survival of the horseshoe crab can be attributed to its inherent adaptability, as well as modern-day government protection. Prior to 1991, there were no laws or regulations in South Carolina pertaining to horseshoe crab conservation. Increased fishing industry demands on the horseshoe crab made it necessary to develop a state-wide management plan to conserve this resource. In 1992, Dr. James Cooper (a pioneer in the research and development of the LAL assay), in partnership with Charles River, wrote draft legislation that called for the management and regulation of horseshoe crab fisheries. As a result, the South Carolina state legislature enacted laws to protect the indigenous horseshoe crab population. In South Carolina, horseshoe crabs must be collected by hand and only be used for biomedical applications (LAL production) and marine biological research, not as bait for the eel and whelk industries.
Since these regulations supporting horseshoe crab conservation have been in place, the horseshoe crab population in South Carolina has flourished.

To further aid horseshoe crab conservation efforts, we only contract licensed fishermen to carefully collect horseshoe crabs from the coastal waters, which are then brought to a facility where their blood is drawn in a highly controlled and monitored environment. Today, the horseshoe crab’s blood is still the most sensitive and reliable detector of dangerous Gram-negative bacteria found in anything from flu vaccines to outer space.

Once the crabs are brought into our lab, they are carefully inspected by trained employees who determine the animal’s health and maturity. At the facility, crabs are cleaned, inspected, and prepped to have their blood drawn. About 25% of the crab’s blood is collected in order to obtain their blood cells, or amebocytes, which are similar to human platelets.

After careful collection of a measured amount of blood, the crabs are returned unharmed to their natural habitat in less than 36 hours, and their blood volume rebounds quickly.
Due to rigorous horseshoe crab conservation efforts, in 2019, the Atlantic States Marine Fisheries Commission (ASMFC) Stock Assessments emphasized that the horseshoe crab population in the Southeastern Atlantic coast has remained in good standing for the last several years. Additionally, the Southeast Area Monitoring & Assessment Program (SEAMAP) Coastal Trawl Survey, conducted by the South Carolina Department of Natural Resources, shows a population increase over the last several years. Results from a South Carolina tagging study also demonstrated that bled horseshoe crabs are able to return to spawning beaches in subsequent years. Charles River’s dedication to horseshoe crab conservation efforts has played a vital role in maintaining the population.


The blue blood of Limulus polyphemus has been invaluable during the pandemic
Read More
In partnership with the South Carolina Aquarium’s Holland Lifelong Learning program, Norm Wainwright, Director of R&D at Charles River, discussed why we owe our lives to the horseshoe crab and the significant role they play in our ecosystem.
Read More
Tag teaming with the South Carolina Aquarium to learn more about the horseshoe crab population.
Read More
Since endotoxins can be so toxic, any product or device that encounters bodily fluids is screened by limulus amoebocyte lysate (LAL) reagents. John Dubczak, GM of Charles River Microbial Solutions, explains the importance of conserving the source of this valuable reagent.

Foster Jordan discusses horseshoe crabs, rFC, and what’s at stake for the pharma industry.
Listen to the Podcast
What is the connection between living fossils and a living person? Ocean lover Heather Bring of the Mystic Aquarium speaks about her work and her knowledge of the horseshoe crabs’ role in her medical care.

A Q&A with Mystic Aquarium Scientist-in-Residence and Charles River Senior Director of R&D, Norm Wainwright.
Read More
The biomedical industry and ongoing conservation efforts continue to be a positive attributing factor to horseshoe crab sustainability.
Watch Now
How plastic pollution affects horseshoe crabs, their coastal habitats, and the ocean at large.
Continue Reading
Herodotus may be the first human historian, but to travel farther back in time we need our fossil friends, the ever-evolving horseshoe crab.
Listen to the Podcast
New Jersey Researchers hatch a plan to raise baby horseshoe crabs and then track them in the wild.
Read More
An assessment of the horseshoe crab stock status in four regions: Northeast, New York, Delaware Bay, and Southeast.
Learn More Here
What are the limitations and restrictions, handling requirements, and penalties when collecting horseshoe crabs in South Carolina?
Read More
Marine scientists document LAL industry's conservation of the horseshoe crab.
Read More
With a total of 88 horseshoe crabs counted, this year’s Mystic Aquarium Horseshoe Crab Counting Walk at Napatree Point in Westerly, RI was a huge success.
Read More
Living Fossils and Blue Blood: The Story of the Horseshoe Crab and Human Health.
Watch the Video
This podcast discusses some of the earliest work undertaken in South Carolina to conserve horseshoe crabs and how it benefits us all.
Listen to the Podcast
Biomedical manufacturers and conservationists alike have a vested interest in making sure these invertebrates continue to thrive and flourish. Find out how the species is faring.
Read More
Eureka guest blogger and longtime South Carolina fisherman, Jerry Gault, talks about his moral duty and legal responsibility to handle horseshoe crabs with care.
Read More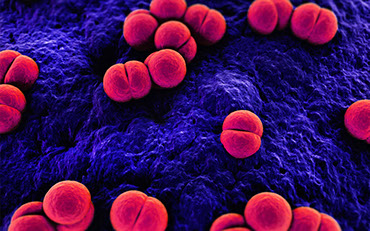
John Dubczak talks about the importance of endotoxin testing in drug development.
Read More
At night, during the full and new moon in late May and early June, the sands of East Coast beaches host a dance that is a half-billion years old.
Read More
Learn about the horseshoe crabs’ remarkable story of survival and adaptability.
Watch the Video
Horseshoe crabs live in the ocean year-round, but they make one annual visit to the shoreline to lay eggs in sandy, wet beaches.
Watch the Video
Find out why the Atlantic horseshoe crab has found allies in the biotech community.
Read MoreReach out for more information about our commitment to Atlantic horseshoe crab conservation or to request a copy of our conservation poster or brochure.